
PathPresenter for Clinical Care
Practical clinical workflow for primary diagnosis in modern institutions
Designed by practicing pathologists and battle-tested in the real world, PathPresenter’s clinical digital pathology software helps professionals do their jobs more quickly and easily. PathPresenter empowers pathologists to receive, view, and sign out cases from their institution – or from remote second opinion cases uploaded from around the world – directly in their institution’s existing LIS, bringing the control and organization of the clinical workflow back to the end user. Our viewer has FDA 510(k) clearance and CE-IVDR certification for primary diagnosis with approved scanners.
Schedule a demo today
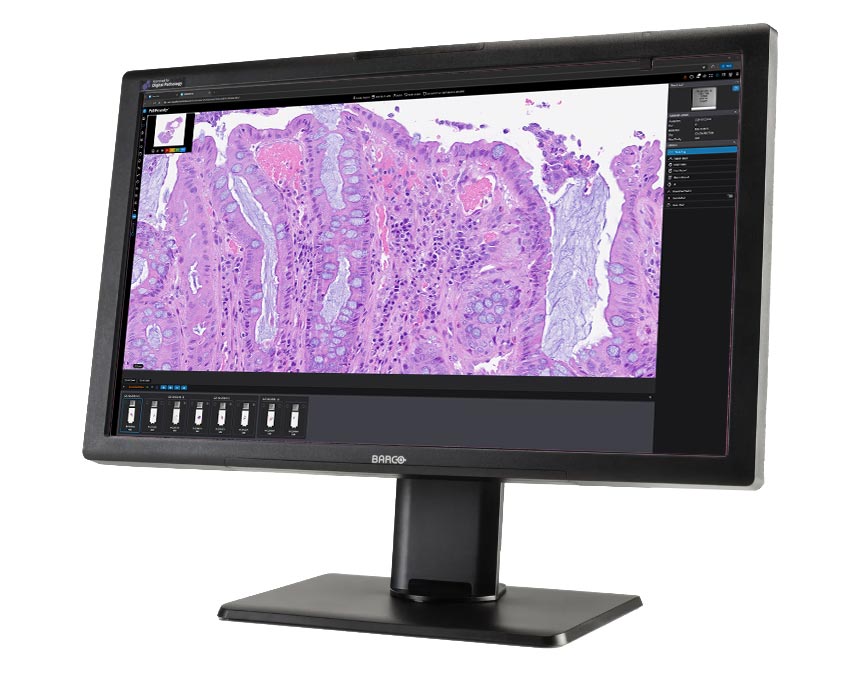
More Speed and Efficiency
PathPresenter’s clinical module is shaped by real-world pathology practice, built in close collaboration with real pathologists who manage high case volumes every day. It’s designed to keep up with the pace of sign-out, even in complex environments with many concurrent users, while maintaining smooth and responsive performance.
Just as importantly, it’s built to make daily work easier and more efficient:
- streamlined workflows that cut down on unnecessary clicks
- flexible preferences that adapt to how you like to work
- macros that reduce repetitive typing
- seamless integration with voice dictation tools
The result is a clinical workflow that is faster, more intuitive, and better aligned with how pathologists actually work.
PathPresenter Receives FDA 510(k) Clearance for Digital Pathology Clinical Viewer
Read more here
PathPresenter Clinical Viewer achieves CE-IVDR certification for Digital Pathology Clinical Viewer
Read more here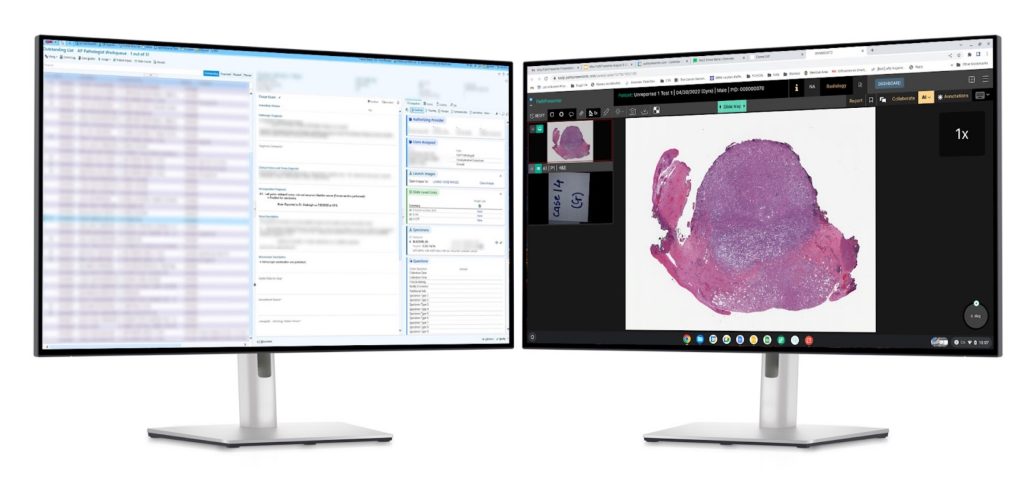
Open slides from your LIS
Sign-out workflow has never been easier. PathPresenter supports deep integration with best in class LIS platforms including EPIC, Oracle Cerner, and LigoLab. A simple mouse click within your existing LIS opens the PathPresenter viewer where the pathologist can view case slides and access best-in-class AI models.

LIS integration through a powerful HL7 middleware layer
Our proprietary HL7 engine supports bidirectional integration that can populate metadata into the LIS. Within the LIS, it creates internal accession numbers that allows images to be linked to cases.

Scanner-agnostic platform
PathPresenter is scanner-agnostic, compatible with all major scanners and file formats, including .svs, .ndpi, .tiff, .mxrs, .bif, .vsi, .jpg, DICOM, multiplex immunohistochemistry/immunofluorescence (mIHC/IF) images, cytopathology z-stack images, as well as gross, clinical and radiology images.

Access best-in-class AI models
In keeping with our “agnostic” system design and emphasis on interoperability, our platform integrates with multiple AI vendors. Pathologists get a consistent UI and workflow and a “future-proof” integration architecture that ensures easy access to the best models in the market today–and tomorrow. PathPresenter is truly your gateway to the best AI models.

Remote Consultations
As a “sharing first’ platform, PathPresenter is designed to connect hospitals and pathologists to other institutions and experts, whether in the same state, country, or around the world. We help institutions expand their remote second opinion programs globally, while removing the significant costs to ship glass slides around the world. Our platform integrates seamlessly into the consulting hospital’s existing infrastructure, making the leap to digital much easier for the pathologist.

Easily Bookmark Interesting Cases for Education or Research
Interesting cases are easily bookmarked and sent into folders for de-identification and use in tumor boards, conferences, research, or education.
The PathPresenter Clinical Viewer is software for digital pathology image viewing and management. In the US, it is has received FDA 510(k) clearance for use in primary diagnosis with the Hamamatsu NanoZoomer S360MD Slide scanner NDPI image formats viewed on the Barco NV MDPC-8127 display device. In the EU it has received CE-IVDR certification with the same configuration. For other regions it has not yet received clearance and is for use in research only.
